
You may have heard that the FDA has come out with new sunscreen guidelines today. While the changes won’t “really” take effect until June 2012, chances are that products with the new labels will start popping up at stores near you in the next 4-6 months.
I’ve previously covered what sunscreens are and how do they work as well as how to pick the right sunscreen for you. Pop on over and read both of those posts if you aren’t familiar with them, I won’t be repeating all of the info in them here.
Basically, SPF currently stands for Sun Protection Factor (the FDA actually admitted today that they toyed with the idea of changing it to Sunburn Protection Factor but decided against this). SPF is multiplied by how long your skin usually takes to burn and how long it will burn instead based on the protection offered by that sunscreen.
• SPF 15 blocks 93% of all incoming UVB rays
• SPF 30 blocks 97% of all incoming UVB rays
• SPF 50 blocks 99% of all incoming UVB rays
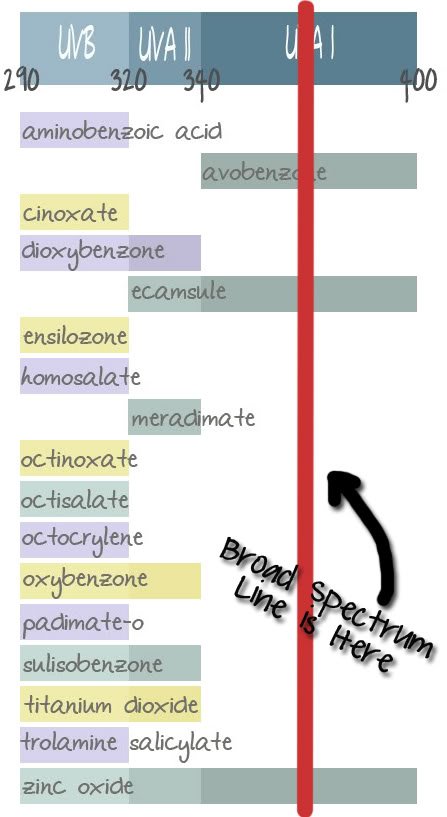
Yes, that’s right. Currently SPF only refers to UVB radiation protection. To protect yourself from UVA radiation you relied on a company using the term “Broad Spectrum” on the label, which was not regulated. One company’s broad spectrum didn’t equal another company’s broad spectrum, since some companies put it only on product that cover short and long wave UVA rays, sometimes only short wave UVA needs to be prevented and I swear I have found it on products that only protect against UVB radiation. What’s broad spectrum in that??
So, US consumers had no way to know how much UVA radiation protection (if any at all) was offered in a product. You may have seen designations such as PA++ on products, but what does this mean and why isn’t it on everything? It is a European designation for UVA protection, but it doesn’t appear on everything in the US. Because it’s… well, meant for Europe. The FDA proposed a 4 star system back in 2007, but nothing had been finalized. Frankly, to me 4 stars seems more appropriate for picking a hotel when I go on vacation, not my sunscreen.
To help readers (and mostly, myself) know if a sunscreen has any UVA protection, I’ve been posting the UV range at the bottom of posts for products that contain sunscreen. So, you can see if there is any short or long wave UVA protection in that product. Unfortunately, I can only tell if the ingredients are there, not how much protection they provide (I’m not cool like Coppertone or Aveeno, I have no at-home sun protection testing capabilities).
More confusion: Water resistant, Very Water Resistant, Waterproof and even Sweatproof. They all appear on sunscreens in the US, despite the FDA saying the “proof” versions shouldn’t be there. And what is the difference between water resistant and very water resistant anyways? (There is a difference, it’s in this post, what makes a sunscreen waterproof.)
New Changes in 2012
So, there are new changes to come, and I’m happy about most of it. Overall, sunscreen labels will provide more information and be easy to read. Score!
SPF
The new SPF will still refer primarily to UVB coverage, though it will sometimes also reflect UVA coverage (see below, broad spectrum, for more info on this). The major difference is that you will no longer see SPF products over 50 on the shelves. Instead they will be labelled 50+. So, those 85, 90 and even 100 products are going bye-bye.
The FDA’s reasoning (and it is correct) is that since SPF 50 already blocks 99% of incoming UVB radiation, studies have found no advantage to using over SPF 50.
My issue is this: Most people don’t use the correct amount of sunscreen. Remember the other day, I told you everyone over at The Pulse Network was shocked when I revealed that the average adult should use 1 ounce (a shot glass) worth of sunscreen? And that your tube is really more like 1 day at the beach, not an entire summer’s worth? Higher SPF sunscreens provide a little more buffer for inadequate sunscreen application. Poor application of a SPF 15 might really give you SPF 8, but poor application of SPF 85 likely puts you over SPF 30.
My second issue: Those of us that are very fair, have a personal or family history of skin cancer, or spend a lot of time outdoors should be using higher SPF value sunscreens. Limiting us to SPF 50 kinda sucks.
I do concede that if you have used your sunscreen appropriately then SPF 50 is likely fine for pretty much everyone.
Broad Spectrum
Remember all of my ranting about the term “broad spectrum” a few paragraphs ago? How it seems to mean something different for most companies? Well, that is a thing of the past.
New rules: SPF will reflect UVB coverage. However, it may also reflect UVA coverage if labelled as “Broad Spectrum”. Basically, the FDA has set it up so SPF is tested to determine the overall UVB coverage number just as before. Then, they test again at 370 nm (in the UVA 1 range). If the coverage passes a certain set point, then the product can be labelled as “Broad Spectrum”. This is a pass/fail test, not an overall percentage. So, if the product is just under the expected set point then it can not be labelled as Broad Spectrum. The best part of this is that the UVA coverage test point changes as the overall SPF changes. So, if you buy an SPF 15 product there is an expected amount of UVA coverage, but the UVA coverage in a SPF 30 product is higher as is the SPF 50+ product.
As you can see from my UV range chart, the new broad spectrum labeling ensures that there will be one of three ingredients in the product: Avobenzone, Ecamsule or Zinc Oxide. My only concern now is that there won’t be UVAII coverage in all products, as you can see it would be possible to formulate a product to have UVB coverage (giving the SPF number) and then adequate coverage in the UVAI range to get the broad spectrum designation without having any coverage in the UVAII range. Because of this I’ll continue to post my UV range charts on sunscreen posts after the changes go into effect next year.
Waterproofing
In theory (?) the terms waterproof and sweatproof weren’t allowed on bottles anymore. Today the FDA even said, “Waterproof wasn’t really allowed, even though we saw this, as well as Sweatproof.” But, you can easily find these terms on some of my favorite sunscreens. I’m hoping that the new rules get rid of these terms completely on bottles, because I have no idea how they were still around.
First, it helps to know about the method of testing water resistance in sunscreens. Human volunteers apply the product to areas of skin that aren’t sun exposed for testing (basically, I think that means their butt?), they swim in an indoor pool for 20 minutes and then air dry. This is repeated multiple times. The SPF that the product tests at after the water exposure is what gets put on the label.
By the old rules, a water resistant product had a total of 40 minutes water exposure, very water resistant products underwent 80 minutes of water exposure. New rules will result in the “Water Resistant” label, but then there will be a time designation- 40 or 80 minutes. So, you know how the product has been tested and when to reapply!
So, I see only good things with this change.
Other Interesting Info
1. New Sunscreens
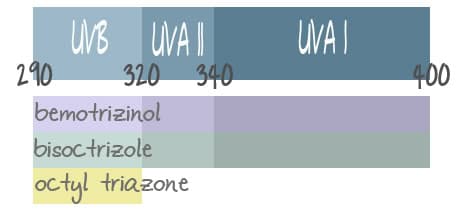
The FDA said in today’s statement that there are other active ingredients that merit investigation into possible approval. This is great news as the US typically lags behind other countries in sunscreen technology. Bemotrizinol (Tinosorb S), bisoctrizole (Tinosorb M), and octyl triazone were all mentioned by name. The Tinosorbs are known to be very photostable. Unfortunately, no timeline for their investigation or possible approval was mentioned.
2. Expiration Dates
You may have noticed that while a 1 year expiration time is usually mentioned for sunscreens, products aren’t labelled to reflect this. The FDA says:
Currently, regulations in 21 CFR 211.137(h) do not require that an expiration date be included in labeling if an OTC drug product does not have any dosage limitations and is stable for at least 3 years. This regulation applies to many OTC drug products, including sunscreen products. Any modification of the existing regulations would require publication of a proposed rule addressing all OTC drug products affected by the expiration date regulations.
So, they won’t require them because it would mean changing a regulation that applies to over the counter medications in addition to sunscreens. Note that while they say it is stable for “at least 3 years”, this typically refers to unopened product. You should still throw away your sunscreen if it has been open for a year.
FDA Press Release
FDA: Labeling and Effectiveness Testing; Sunscreen Drug Products for Over-the-Counter Human Use

 I’m a doctor, a mommy and a bit of a beauty addict. If you let me, I can take 2 hours to get ready in the morning. Really. I'm on a quest for faster beauty that works!
I’m a doctor, a mommy and a bit of a beauty addict. If you let me, I can take 2 hours to get ready in the morning. Really. I'm on a quest for faster beauty that works!
It's about time they got rid of all that SPF 100 malarky – anything about SPF 30 is really just going into the laws of diminishing returns. Applied properly, and with a physical sunscreen (rather than chemical) even the very fair should have all the protection they need.
This is a great website I found on sunscreens http://breakingnews.ewg.org/2011sunscreen/ and I no longer purchase without consulting this first! 🙂
I disagree about diminishing returns over SPF 30. For those with fair skin that burns easily (me), there is a noticeable difference between SPF 30 and 50. I'm the poster girl for proper application so the difference is not using enough sunscreen. As well, only a very small percentage of people use the proper amount of sunscreen per application, so the extra "buffer" of a higher SPF can be very important. Sunscreens SPF 50 and higher have been recommended for those with a personal history of skin cancer, SPF 30 doesn't fit everyone.
As well, you should be aware that the Environmental Working Group is a political action group, with a strong stance against any chemicals. As a lobbyist group, they are not an impartial source of information and have been criticized for incorrectly drawing conclusions from studies by many physicians and scientists. This is fine if you agree with their stance, but just keep in this in mind.
This is such a great post. Very informative!
I want to thank you for your series on SunScreens. I am reading them all (and bookmarking some). You have given good explanations and your UVA/UVB chart is very clear and helpful.
I am one of those SPF +90 and above users. I can't spend an hour at the pool or at the beach without slathering that SPF rating on. I start burning around 20 minutes without it.
And I don't think that I use the right amount. A tube will last me a week. But too much of that can make my face break out—not acne, red bumps and welts.
I suppose that I will have to stock up on the SPF +90.
Ecamsule is Mexoryl SX? So has Mexoryl XL been approved for the US? And I should start looking for Tinosorbs on the internet drugstores? Who makes a brand with that in it?
I swear, I have to bring a Cheat Sheet with me when I am buying SPF. 8-p
You've done a great job explaining a heady subject. Your wavelength absorption graphic is so helpful for clarifying the specifics.
As you know I'm a mineral sunscreen gal and love my zinc ox. The mechanism of action as a stable reflector is what gives me such confidence. It's also much less likely to irritate sensitive skin (aka mine!).
Great reiteration of the 1 oz per adult body per application point too! I love to tell people that a tube is 1 day at the beach…..so you might want to coverup some of the skin surface w/ sun protective clothing, it's less work and probably less expensive in the long run. It's also better protection since you're less likely to miss spots or application intervals.
Here's the 'trouble' I go through when I'm in extreme sun http://www.drbaileyskincare.com/blog/savvy-sun-protection-for-your-winter-getaway-lessons-from-my-polynesian-cruise/ It works and helps prevent wrinkles, age spots, cancers and the like.
Great job!
Cynthia Bailey MD, Dermatologist
I'm glad to see the FDA is going to be making companies prove some of their ridiculous claims and whatnot. Maybe if everyone is worried about all the people who don't use sunscreen correctly (though, if they're not applying the correct amount, they're probably also not applying it everywhere it needs to go or as often as they should, etc., so I don't think a decrease in potential sun protection will make a huge difference to most people), the FDA or sunscreen companies should fund some PSAs. I'm sure the sunscreen companies could only benefit by telling everyone how much sunscreen they should be using lol
Chatchien-
I'm happy you've found my posts so helpful! In regards to the problems you've had with sunscreens on your face, have you tried a completely physical sunscreen on your face? The problems you are having sounds like a bad reaction, and I wonder if a non-chemical option would be better.
Ecamsule is Mexoryl SX, I don't know that XL has actually been approved in the US as it is a different chemical and that name doesn't appear on the approved list here in the US. There's a good explanation of Mexoryl here (they have good info on lots of chemicals on this site, I just found it in Google searching to figure out the name of mexoryl XL), and on this site one of the tinosorbs in a physical sunscreen and the other is a hybrid chemical/physical. They aren't approved in the US, so I'm not sure who makes them for Europe. I would bet that Lumene and La Roche Posay ones are available, those are the sunscreen brands I always hear that people stock up on when they visit Europe.
I have a saved copy of my sunscreen ingredients graphic on my iPhone for when I'm shopping. So yes, I really do have a cheat sheet! I think the new FDA guidelines will help eliminate most of that. We'll just have to look for over SPF 15 and broad spectrum on the package.
Cynthia- Thanks so much for your nice comments! I was really curious what you would think of this post. I really agree about the sunsmart clothing. I've been shopping for some recently since my 4 month old (who seems to be a pale strawberry blonde, if I can figure it out from her short baby fine hair) is too young for sunscreens. If you have favorite brands of sun smart clothing I'd love to know who they are!
Ok yeah this was all very informative, so thank you very much. But it gets very confusing trying to remember all that when buying a sunblock/sunscreen, then piling on loads and loads of sunscreen everyday, I don't know where to start.
Bere-
Start here: Sunscreen 101: How to Pick the Right One for You, and when you know kind of what you are looking for shoot me an email at cemikese at yahoo.com and I'll help you from there. 😀
I completely agree with you that SPF greater than 30 is needed for us pale girls. What can it hurt to use SPF 100 if it's the same price as SPF 30, I need all the help I can get.
This is a great post about the new sunscreen regulations! I was going to write about it on my blog but maybe I will just link to your post instead! I always wear zinc oxide-based sunscreens, and I'm curious to see if it will become a much more commonly used ingredient now.
Wow! Thank you so much for such an informative post. This will definitly help come this summer when my work commute is going to be significantly longer. (I seem to always burn on car rides)
Hi! Thank you for all your informative posts! I was wondering if the new sunscreens that included Tinosorb or Uvinul are safe for use while pregnant? Zinc oxide sunscreens seem to burn my skin over time. Thank you for your help! 🙂
Tinosorb and Uvinul are both chemical sunscreens and should be avoided in pregnancy.