Sunscreen is one of those must have products that just isn’t terribly sexy. A new lash serum? A great face peel? What about the latest eye shadow palette? All of those are a lot of fun. But sunscreen is a basic product that you should be using every day.
Unfortunately, sunscreen isn’t super straight forward! Not all products are created equal, and you’ll need to know what the label needs when you’re shopping for a new sunscreen. Note that most products are reformulated yearly or bi-yearly.
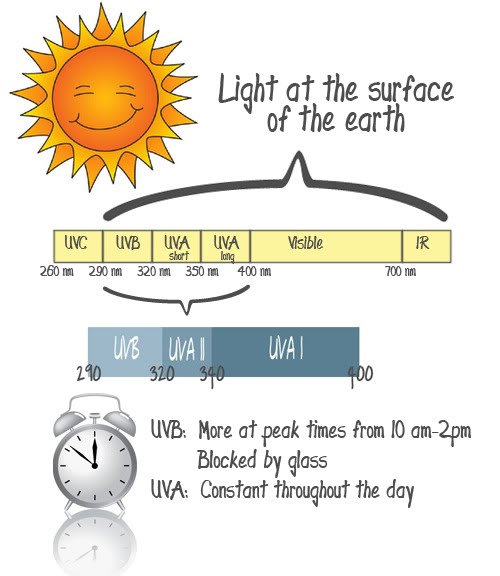
Types of Sun Rays
So, what’s the deal with all the different types of UV radiation? It’s all emited by the sun, but that doesn’t mean that it affects us. UVC radiation actually never even reaches the earth’s surface, it’s all filtered out by our atmosphere. So, any UVC exposure we get is all from artificial sources. We’re going to chose to ignore the visible spectrum and the infrared (IR) spectrum since we can’t protect against it and I’m not even sure if anyone knows much about it’s affects on the skin (but, that doesn’t mean we should totally ignore it in our real life, that’s what sun exposure avoidance is for. I’m just choosing not to talk about it in this post). We’re worried about UVA and UVB exposure here.
When sunscreens were first developed they were against UVB rays, since this is what causes the redness we associate with a sun burn. So, what do each of these ranges really do to skin?
• UVB: Skin redness and burning
• UVA: Lowers skin immunity, increasing cancer risk, penetrates to the dermis, contributes to aging via increased skin thickness and increased skin inflammation, tanning of the skin
• Both: Induces DNA damage in skin, increasing cancer risk
What is SPF?
SPF stands for Sun Protection Factor, and yes, a SPF really is multiplied by how long your skin usually takes to burn. So, if you usually take 10 minutes to burn, with well applied SPF 30 that equals 300 minutes.
I recently found another way to think about it on The Skin Cancer Foundation website:
• SPF 15 blocks 93% of all incoming UVB rays
• SPF 30 blocks 97% of all incoming UVB rays
• SPF 50 blocks 99% of all incoming UVB rays
How do Sunscreens Work?
There are two main types of sunscreens, physical and chemical.
• Physical: These sunscreens really work by blocking the light from ever reaching your skin. The light is reflected or scattered away from the skin. These ingredients are less likely to irritate skin or create an allergic reaction. However, they tend to create a white cast on the skin, contribute to a product having that “sunscreen” feeling, and they can stain clothing.
• Chemical: These sunscreens absorb UV radiation rather than reflecting it away from skin. To do this, the energy of the light has to go somewhere, or do something. Depending on the ingredient, the light can be used in a chemical reaction, can slightly alter the chemical structure of the sunscreen itself, be given off as heat (I’m not sure that any sunscreens approved in the US actually do this), or be released as a longer UV wavelength. While these chemicals don’t feel as heavy as their physical sunscreen counterparts and can cover a wide range of UV wavelengths, they also have their drawbacks.
Chemical sunscreens can be unstable in certain combinations or with prolonged sun exposure (hence we docs tell you to reapply often). They are implicated more often in skin irritation and allergic reactions. And they can even be absorbed into the skin, and picked up in your urine.
Note: The FDA considers Ecamsule (Mexoryl) to only be a UVAII screen, however all of the data I found stated that it does in fact screen out UVAI as well, but not super well. Hence, I have altered my own charts to reflect this.
What SPF Level Do You Need?
Ideally, this is a higher the better situation. You should use the highest SPF you feel comfortable wearing every day. Buying SPF 55 isn’t ideal if you can’t stand to put it on your skin, so you either skip using the product completely or only about half of the recommended amount.
• Most dermatologists recommend SPF 30 and should be fine for your normal “out and about” life if you are running errands, going to work, et cetera. If you are spending any significant amount of time outside, you likely need a higher SPF.
• A lot of moisturizers and foundations contain SPF 15. I feel very strongly that you need to think about what that product is and how much of it you apply before you decide if that is “enough” SPF for you. If it is foundation and you only apply it in your t-zone, it is not going to protect your cheeks. If it is in your moisturizer which you smear all over your face, neck and chest (hint: you should be doing this already), then you probably are ok.
• If you are prone to sunburns or have a personal/family history of skin cancer, you really should use a higher SPF. A lot of people say “oh, there isn’t a big difference between SPF 30 and 50” but that difference in 97% vs 99% UVB blockage can make a big difference if you have a family history.
You can check out my post on The Secret to Picking The Right Sunscreen!
Application Tips
• The quoted SPF is based on ideal application. That means at least an ounce of sunscreen for your body, a rule followed pretty much only by me and dermatologists.
• Even if you have followed all of the rules, you need to reapply after 2 hours. Why? Regardless of all the “time” you have for it to work, no sunscreen is meant to work for more than 2 hours. In fact, there is research that shows sunscreen that has been on for too long is counterproductive and (if I’m remembering correctly, I can’t find the study right now) actually contributes to skin damage.
• I don’t care if you are wearing a water resistant formula, no matter what you need to reapply that sunscreen as soon as you climb out of the pool.
• Don’t forget that ideal sunscreen application takes place 30 minutes prior to sun exposure, allowing it to bind with your skin. That means not at the pool side and not while your kids are running around the backyard already. Reapplication there is ok, but the best initial application is inside. I usually do this even before I put on my bathing suit, which helps to prevent missed areas next to the edge, and I can much more easily contort myself in the hotel bathroom than next to the pool.
Moral of the story? Wear a high SPF, apply a lot and reapply often!
Full Spectrum Sunscreen
Why Full Spectrum Sunscreen is Better Than Broad Spectrum
Full Spectrum Body Sunscreens
8 Full Spectrum Facial Sunscreens
Pregnancy Safe Full Spectrum Sunscreens
Pregnancy Safe Full Spectrum Facial Sunscreens, BB & CC Creams
Suggested posts on sunscreen
Dermatologist Sunscreen Tips from Dr. Elizabeth Hale
Not Just Sunscreen: Dermatologist Tips For Out In the Sun
Mistakes you’re making with your sunscreen
The Secret to Picking The Right Sunscreen!
How to Reapply Sunscreen (While Wearing Makeup)
How to Correctly Apply Sunscreen
How to Treat a Sunburn